Stanway J, Birrell F. Treating to target will optimise long-term outcomes in RA. Practitioner November 2020;264(1842):17-20
Treating to target will optimise long-term outcomes in RA
24 Nov 2020
AUTHORS
Dr James Stanway MB BS MRCP Academic Specialist Registrar in Rheumatology
Dr Fraser Birrell MB BChir MA PhD FRCP PFHEA, Consultant and Honorary Clinical Senior Lecturer in Rheumatology, Northumbria Healthcare NHS Foundation Trust, Newcastle University, Newcastle upon Tyne, UK
Competing interests: None
Article
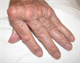
Rheumatoid arthritis (RA) classically causes a symmetrical, small joint polyarthritis which left untreated can lead to joint destruction and deformity. Patients typically present with stiffness, pain and swelling of the small joints in the hands and wrists which may lead to functional impairment. However, RA can affect any synovial joints, apart from sparing the distal interphalangeal joints. Prevalence is around 1%, and women are more commonly affected than men, the median age of onset is 58 years. Younger patients are more likely to be positive to anti-CCP and more likely to present with classical, symmetrical, small joint polyarthritis. The key aspect of assessment in primary care is to ascertain if a patient’s musculoskeletal symptoms are inflammatory or non-inflammatory. Cardinal features that suggest inflammation are: prolonged or severe early morning stiffness lasting > 1 hour; episodic symptoms; joint swelling and improvement of symptoms on exercising the affected joints. RA is a clinical diagnosis, based on history, examination, blood tests and imaging. NICE recommends that any adult with suspected persistent synovitis of undetermined cause should be referred for a specialist opinion. Patients should be referred urgently (even with a normal acute-phase response, negative anti-CCP antibodies or rheumatoid factor) if any of the following apply: the small joints of the hands or feet are affected; > 1 joint is affected; there has been a delay of ≥ 3 months between onset of symptoms and seeking medical advice. Classification criteria for RA consist of four domains: pattern of joint involvement; duration of symptoms; inflammatory markers and serology. Early commencement of DMARD therapy is recommended for both younger and older patients unless there are contraindications. A treat to target approach is recommended.
Rheumatoid arthritis (RA) classically causes a symmetrical, small joint polyarthritis which left untreated can lead to joint destruction and deformity.
Patients typically present with stiffness, pain and swelling of the small joints in the hands and wrists (metacarpophalangeal and proximal interphalangeal joints) which may lead to functional impairment. However, RA can affect any synovial joints, apart from sparing the distal interphalangeal joints.
RA is relatively common, with a prevalence of around 1%, and women are more commonly affected than men.1 Median age of onset is 58 years.2 There is no universally agreed age classification in RA, some classify over 60 as late onset whereas others have used the median age as a cut-off between late onset and young onset.2,3
Clinical features differ somewhat between the young and late onset groups. Younger patients are more likely to be positive to anti-citrullinated peptide antibody (ACPA/anti-CCP); more likely to present with classical, symmetrical, small joint polyarthritis and the female predominance is more pronounced.
It is not clear if one group has a more severe disease course than the other. Answering this question is complicated by the fact that young patients are significantly more likely to be commenced on disease modifying anti-rheumatic drugs (DMARDs), despite the fact that current guidelines do not make differing recommendations based on age and early DMARD initiation is recommended for both younger and older patients unless contraindicated on clinical grounds.
An important contrast should be drawn with juvenile idiopathic arthritis (JIA); an individual developing persistent joint inflammation under the age of 16 is still classified as having JIA when the inflammation persists into adulthood.
Regardless of age, the key aspect of assessment in primary care is to ascertain if a patient’s musculoskeletal symptoms are inflammatory or non-inflammatory. Although distinguishing between the two can be difficult on clinical grounds alone and requires ultrasound imaging in some cases, the cardinal features that suggest inflammation are:
• Prolonged or severe early morning stiffness lasting longer than an hour
• Episodic nature of symptoms
• Presence of joint swelling
• Noticeable improvement in symptoms on exercising the affected joints
All cases of suspected inflammatory arthritis (with the exception of podagra, where a diagnosis of gout is clear) warrant assessment by a rheumatologist, first to confirm the presence of joint inflammation and second to classify the type of inflammatory arthritis.
RA is the most common form of chronic inflammatory arthritis. Although the majority of cases (70-80%) are seropositive, 20-30% are seronegative. The differential diagnoses include:
• Seronegative arthritides (mnemonic ‘PEAR’)
Psoriatic arthritis
Enteropathic arthritis (e.g. associated with Crohn’s disease or ulcerative colitis)
Axial spondyloarthritis
Reactive arthritis (from genitourinary or gastrointestinal infection: both can also be associated with conjunctivitis and urethritis)
• Crystal arthritis (gout and pseudogout)
• Septic arthritis
• Connective tissue diseases such as SLE
• Sarcoidosis
• Paraneoplastic arthritis
• Viral infection (classically parvovirus, but many other viruses can trigger this and viruses can also trigger reactive arthritis)
Given the wide differential, it is important to look for the presence of associated conditions such as psoriasis (where a family history and the presence of nail psoriasis are key features sometimes missed), inflammatory bowel disease as well as symptoms suggestive of multisystem connective tissue disease. Crystal arthritis is an important differential and pseudogout can mimic RA though this is rare in younger patients. Gout is exceedingly rare in premenopausal women, but an important differential in younger men, especially with a family history and other risk factors such as high alcohol intake.
Another important presentation is seropositive arthralgia. This describes a group of patients with positive ACPA and joint pains but lacking clinical evidence of joint inflammation precluding a diagnosis of RA. This group are at high risk of progression to RA and still warrant assessment by a rheumatologist. The role of DMARDs in this group to prevent the development of RA is the subject of ongoing research.
Confirming diagnosis
RA is a clinical diagnosis, based on history, examination, blood tests and imaging (see table 1). Initial tests should include full blood count; routine biochemistry; ESR and CRP; rheumatoid factor (RF); anti-CCP and anti-nuclear antibodies (ANA) and antibodies to extractable nuclear antigens (ENAs) as well as serum urate.
Rheumatoid factor (RF) is a non-specific test and despite its name actually has a greater sensitivity for Sjögren’s syndrome rather than RA (where Ro & La antibodies may be positive, CRP is typically normal but the ESR elevated secondary to the polyclonal increase in immunoglobulins, which accentuates rouleaux formation). Other causes of elevated RF include connective tissue diseases, chronic infections and bacterial endocarditis. Anti-CCP antibodies are of greater diagnostic value with a significantly greater specificity for RA.4
The NICE recommendations on referral to a specialist are listed in table 2.
The current classification criteria for RA were published by the American College of Rheumatology (ACR) and the European League Against Rheumatism (EULAR) in 2010 (see table 3) and consist of four domains:5
• Pattern of joint involvement
• Duration of symptoms
• Inflammatory markers
• Serology
Six points from any of the four domains are required to make a diagnosis. Note that autoantibodies are not a prerequisite for diagnosis and, as mentioned earlier, around 20-30% of RA cases are seronegative. Seronegative RA tends to have a less severe course than seropositive disease but the current treatment strategies are identical.6
Imaging should be arranged at baseline and include plain radiographs of hands, feet and chest. Although a chest X-ray does not aid the diagnosis of RA, it helps rule out paraneoplastic presentations and sarcoidosis. A chest X-ray is also an important screening test for respiratory pathology which may impact on the decision to use methotrexate. Many centres would also perform pulmonary function tests to establish a baseline for comparison on methotrexate.
Where available, specialists often use ultrasound imaging to provide an objective measure of joint inflammation, especially if clinically uncertain or if the diagnosis is unclear, or for guided injection of inaccessible joints such as the hip. This is particularly useful in patients with an elevated BMI where clinical examination for synovitis can be challenging and the presence of low-grade elevation in inflammatory markers is of unclear significance, as fatty tissue directly releases CRP, requiring the normal range to be adjusted.
Ultrasound may also be useful in monitoring response to treatment in patients for whom it is not clear if their symptoms are a result of ongoing, active inflammation or a result of long-term joint damage.
Assessing disease severity
Clinically, disease activity is monitored by the DAS-28 score, a composite scoring tool comprising four features: swollen joint count (0-28); tender joint count (0-28); patient reported severity on a visual analogue scale (0-10) and the level of inflammatory markers (either ESR or CRP).
The DAS-28 score can be calculated using online or app-based tools and produces a numerical value which can be categorised as remission, mild, moderate or high disease activity (see table 4).
The presence of bony erosions on plain X-rays is also a useful marker of poor prognosis and is associated with a greater degree of functional impairment.7
Management
Once the diagnosis is confirmed, DMARDs should be discussed with the patient including thorough counselling regarding the risks, side effects and monitoring requirements; DMARD selection should be a joint decision between the patient and rheumatologist. As mentioned above, early commencement of DMARD therapy is recommended for both younger and older patients unless there are contraindications.
Methotrexate is recommended as the first-line conventional synthetic DMARD (csDMARD), subcutaneous administration is preferable to oral, and higher doses may be achieved.8 Corticosteroids are also advised as bridging therapy as csDMARDs may take up to three months to reach their full effect.
A treat to target approach is recommended, with the goal of treatment to achieve at least low disease activity (DAS-28 < 3.2), although the treatment target should be agreed with each individual patient and in younger patients with high-risk features (ACPA-positive, raised inflammatory markers and erosions) a target of remission (DAS-28 < 2.6) may be more suitable.
Damage to joints and the cardiovascular system is driven by the area under the curve of inflammation; both the magnitude of CRP or ESR and time they are elevated predicts damage, so high and prolonged inflammation interact with other cardiovascular risk factors. We would recommend the use of a cardiovascular risk score such as QRISK3, which includes RA.
Following diagnosis, DAS-28 should be assessed on a regular basis (every 1-3 months) to ensure an adequate treatment response.
If there has been insufficient improvement following DMARD initiation, a second csDMARD is recommended in addition. Further corticosteroid therapy may also be required.9
Current NICE guidelines state that if high disease activity (DAS-28 > 5.1) persists despite two csDMARDS, then treatment with a biologic (bDMARD) or targeted synthetic (tsDMARD) should be offered.10 At present, the first-line bDMARD is generally a TNF-alpha inhibitor (such as adalimumab, etanercept, certolizumab ). Should the first anti-TNF not succeed in achieving adequate disease control, a second
anti-TNF may be trialled or a different pathway could be targeted. Current options include B-cell depletion (rituximab); inhibition of T-cell activation (abatacept) and inhibitors of IL-6 signalling (tocilizumab and sarilumab).
The JAK inhibitors (baricitinib and tofacitinib) are the most recent additions to the treatment of RA. In contrast to biologics, these tsDMARDs are small molecule drugs that can be taken orally and are considerably cheaper to produce.
Two important considerations with all biologic therapies are immune suppression and occasional counter-intuitive induction of autoimmune disease. The former means that the signs and symptoms of infection can be less and both the patient and primary care team need to have a high index of suspicion for infection and low threshold for contacting the secondary care team to rule out infection, omitting the biologic therapy until confident that infection has been ruled out.
A particular issue with IL-6 and JAK inhibitors, is their ability to suppress the production of acute phase reactants such as CRP, without entirely suppressing inflammation. Not only can this give a false impression of efficacy when measuring disease activity using the DAS-28 score, it can also complicate the diagnosis of infection.
The two key issues with treating young adults of child-bearing age are the effects of active disease vs DMARD treatment on conception and teratogenic risk.
There is evidence that fertility is not affected in either men or women, apart from a well recognised effect of sulfalsalazine temporarily reducing sperm count.11 However, high disease activity before pregnancy may have an important contribution to both the time to conceive and worse pregnancy outcome.12
It is well established that antimetabolites such as methotrexate and leflunomide are contraindicated (see table 5). Methotrexate should be stopped at least 3 months prior to conception, whereas leflunomide would need to be actively cleared from the body given its enterohepatic circulation and extended half-life. In contrast, sulfasalazine and hydroxychloroquine are both safe to continue during pregnancy and when breast-feeding. Many patients find disease activity improves during pregnancy, so shared treatment decisions and personalised care are important in the period before, during and after pregnancy.
Because of its molecular structure, the anti-TNF agent certolizumab does not cross the placenta and is considered the safest biologic option during pregnancy.13,14
Conclusion
RA is a lifelong condition with the potential to cause significant morbidity and increased mortality, especially in those with multiple cardiovascular risk factors.
Early assessment by a rheumatologist is key and the prompt initiation of DMARDs vital. Methotrexate remains the mainstay of treatment, with evidence that subcutaneous methotrexate is better tolerated than oral though a range of more targeted options are available.
In the future, it is hoped that therapies designed to reverse the underlying autoimmunity could prevent and perhaps even cure RA.15
REFERENCES
1 Symmons D, Turner G, Webb R et al. The prevalence of rheumatoid arthritis in the United Kingdom: new estimates for a new century. Rheumatology 2002;41;793-800
2 Innala L, Berglin E, Möller B et al. Age at onset determines severity and choice of treatment in early rheumatoid arthritis: a prospective study. Arthritis Res Ther 2014;16:R94
3 van Schaardenburg D, Breedveld, FC. Elderly-onset rheumatoid arthritis. Sem Arthritis Rheum 1994;23:367-78
4 Song YW, Kang EH. Autoantibodies in rheumatoid arthritis: rheumatoid factors and anticitrullinated protein antibodies. QJM Int J Med 2010;103:139-46
5 Aletaha D, Neogi T, Silman AJ et al. 2010 Rheumatoid arthritis classification criteria: an American College of Rheumatology/ European League Against Rheumatism collaborative initiative. Ann Rheum Dis 2010;69:1580-88
6 Kroot E-JJA, Jong BAWD, Leeuwen MAV et al. The prognostic value of anti–cyclic citrullinated peptide antibody in patients with recent-onset rheumatoid arthritis. Arthritis Rheum 2000;43:1831-35
7 Ødegård S, Landewé R, van der Heijde D et al. Association of early radiographic damage with impaired physical function in rheumatoid arthritis: a ten-year, longitudinal observational study in 238 patients. Arthritis Rheum 2006;54:68-75
8 Li CK, Baker K, Jones T et al. Safety and tolerability of subcutaneous methotrexate in routine clinical practice. Arthritis Care Res 2020 (advanced access) doi:10.1002/acr.24334
9 Smolen JS, Landewé RBM, Bijlsma JWJ et al. EULAR recommendations for the management of rheumatoid arthritis with synthetic and biological disease-modifying antirheumatic drugs: 2019 update. Ann Rheum Dis 2020;79:685-99
10 National Institute for Health and Care Excellence. NG100. Rheumatoid arthritis in adults: management. NICE. London. 2018 updated 2020 [Last accessed 10 Nov 2020]
11 De Cock D, Brants L, Soenen I et al. A systematic review on the effect of DMARDs on fertility in rheumatoid arthritis. Semin Arthritis Rheum 2020;50:873-78
12 de Man YA, Dolhain RJEM, Hazes JMW. Disease activity or remission of rheumatoid arthritis before, during and following pregnancy. Cur Opin Rheumatol 2014;26:329-33
13 Clowse MEB, Scheuerle AE, Chambers C et al. Pregnancy outcomes after exposure to certolizumab pegol. Arthritis Rheumatol 2018;70:1399-1407
14 Krause ML, Makol A. Management of rheumatoid arthritis during pregnancy: challenges and solutions. Rheumatol Res Rev 2016;8:23-36
15 Stanway JA, Isaacs JD. Tolerance-inducing medicines in autoimmunity: rheumatology and beyond. Lancet Rheumatol 2020;2:e565-75
ARTICLE IN PDF
EXTERNAL WEBLINKS
National Rheumatoid Arthritis Society
British Society for Rheumatology
British Society of Lifestyle Medicine
Arthritis and musculoskeletal virtual group consultation resources

 = Registered users
= Registered users = Paid-up subscribers
= Paid-up subscribers